thank you very much for thisprecious information.... after i excluded allproducts that contain this ingredient, my severely deep inflammed cystic acne were cleared up significantly
26種被歐盟規範禁用的化妝品添加香料。
http://maskingdom.pixnet.net/blog/post/151084026-26%E7%A8%AE%E6%AD%90%E7%9B%9F%E8%A6%8F%E7%AF%84%E7%9A%84%E5%8D%B1%E9%9A%AA%E9%A6%99%E6%96%99%EF%BC%81
Possible human immune system toxicant or allergen
https://www.ewg.org/skindeep/ingredient/700872/LILIAL/
Safety
The safety of butylphenyl methylpropional has been evaluated by the Research Institute for Fragrance Materials Expert Panel (REXPAN). Based on this evaluation, an International Fragrance Association (IFRA) Standard has been established. The IFRA Standard restricts the use of butylphenyl methylpropional in fragrances because of potential dermal sensitization.
A research article published in the Flavour and Fragrance Journal assessed the risk of sensitization for 26 fragrances in 5451 products from 2007 to 2009. As an estimate of sensitization risk, the sensitization exposure quotient (SEQ), was calculated as the quotient of the relative frequency of sensitization and the relative frequency of use/labelling. The SEQs (the risk) varied greatly, with butylphenyl methylpropional at the bottom of the list, indicating a very low risk of sensitization.
Since there is risk for sensitization with butylphenyl methylpropional, its presence must be indicated in the list of ingredients if the product contains more than 0.001% and is meant to be left on skin. It also must be listed if the product contains more than 0.01% and is meant to be rinsed, such as with cleansers and shampoos.
Due to the possibility of an allergic reaction, those with sensitive skin should try to butylphenyl methylpropional or perform a patch test with any product containing this ingredient.
References: Wikipedia, “Lillial”, Flavour Fragr. J., 2015, 30: 208–217, Cosmetics Info, “Butylphenyl methylpropional”, European Commission Scientific Committee on Consumer Safety “OPINION ON Butylphenyl methylpropional (BMHCA)”, 2016.
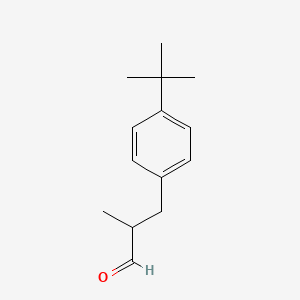
Butylphenyl Methylpropional
丁苯基甲基丙醛
丁苯基甲基丙醛在化妆品、护肤品里主要作用是抗氧化剂,香精香料,风险系数为7,要多留意反应,对于孕妇一般没有影响,丁苯基甲基丙醛没有致痘性。
可抑制皮肤表层细胞新陈代谢的速度,延缓细胞衰老,过量时,具有毒害作用。
參考資料:https://m.kqmmm.com/cosmetic/201184.html
法令新知 |歐盟對香精香水類的新法規(EC) No 1272/2008
發佈時間:2022/02/10
[本新聞已曾於2021/04/29即在本欄上架,明年即將生效,今重新上架提醒化妝品輸歐企業]
丁苯基甲基丙醛Butylphenyl Methylpropional,俗稱Lilial,具鈴蘭或百合花氣味,被歸類為過敏原及致癌物CMR 1B等級,此化合物存在於眾多化妝品中的香精成分,該法案增修原法條附件六(Annex VI)的第三部分,包括相關分類、標示與包裝的規範,且包括該物質本身及其混合物。
歐盟化妝品法規 EC 1223/2009禁止使用被歸類為致癌物的成分物質(CMR,致癌/致畸胎/致突變),除非列於第15款的免除名單者除外。此一有關Lilial的新分類將於2022/3/1生效,何時禁用尚未公告,但我們建議產品有使用的企業,盡早啟動取代Lilial的香精成分。
參考資料:https://www.e-sinew.com/shownews.php?storyid=1363
【Butylphenyl Methylpropional(丁苯基甲基丙醛)】—》歐盟已禁用!
Rating: WORST
Categories: Fragrance: Synthetic and Natural, Irritant
Butylphenyl methylpropional is a synthetic fragrance ingredient with a strong floral scent. It also goes by the name 2-(4-tert-butylbenzyl) propionaldehyde or lilial. Butylphenyl methylpropional’s use in cosmetics is restricted due to concerns over sensitivity, especially if multiple products (both personal care and household items such as air freshener) are used routinely.
This ingredient is banned for use in cosmetic and personal care products sold in European Union member countries due to potential reproductive harms in people. Worth noting is that this ingredient rarely shows up in skin care or makeup products, being more common as a fragrance additive in hair care products, perfumes and household cleansers.
In countries where butylphenyl methylpropional is still permitted for use, it must be indicated on the ingredient list if the product contains more than 0.001% and is meant to be left on skin; it also must be listed if the product contains more than 0.01% and is meant to be rinsed. This would not apply to its usage in household cleaners and laundry products.
https://www.paulaschoice.com/ingredient-dictionary/ingredient-butylphenyl-methylpropional.html
